Takeaway And Helpful Resources
The dosages in this article are typical dosages provided by the drug manufacturer. If your doctor recommends Cosentyx for you, theyll prescribe the dosage thats right for you. Always follow the dosage that your doctor prescribes for you.
As with any drug, never change your dosage of Cosentyx without your doctors recommendation. If you have questions about the dosage of Cosentyx thats right for you, talk with your doctor.
Besides learning about dosage, you may want other information about Cosentyx. These additional articles might be helpful to you:
- More about Cosentyx. For information about other aspects of Cosentyx, refer to this article.
- Side effects. To learn about side effects of Cosentyx, see the Cosentyx medication guide.
- Drug comparison. To find out how Cosentyx compares with Stelara, read this article. And to learn how Cosentyx compares with Humira, read this article.
- Details on your condition. For details on your condition, see our lists of:
Dosing Of Subcutaneous Non
Product comparisons with regard to efficacy and safety cannot be made in the absence of head-to-head clinical studies. This presentation is not intended to compare the relative safety or efficacy of these treatments. Please refer to the full Prescribing Information of each agent for dosage and administration.
Taltz is a registered trademark owned or licensed by Eli Lilly and Company, its subsidiaries, or affiliates.
Cosentyx is a registered trademark of Novartis AG.
*Four injections per yearonce every 12 weeks after starter injections, in the first year, at Weeks 0 and 4.
In a patient > 220 lb, the recommended dose for STELARA® is 90 mg initially and 4 weeks later, followed by 90 mg every 12 weeks. In this case, the number of injections per year would be 12.
Total number of injections in the first 52 weeks of therapy.
§For patients with PsA with coexistent moderate to severe plaque psoriasis, use the dosing and administration recommendations for plaque psoriasis. For other patients with PsA, administer Cosentyx® with or without a loading dose by subQ injection. The recommended dosage:
- With a loading dose is 150 mg at Weeks 0, 1, 2, 3, and 4 and every 4 weeks thereafter
- Without a loading dose is 150 mg every 4 weeks
- If a patient continues to have active PsA, consider a dosage of 300 mg every 4 weeks. Cosentyx® may be administered with or without MTX
Product Indications
Taltz® is indicated for the treatment of adults with active PsA.3
How Is Cosentyx Used
Cosentyx is a medication thats injected under your skin. You can find helpful videos about how to inject Cosentyx on the manufacturers website. You can also talk with a Cosentyx support specialist and get live virtual injection training. Call Cosentyx Connect at 844-COSENTYX for this service.
You can also ask your pharmacist for more information on how to inject Cosentyx.
Also Check: Severe Psoriasis And Psoriatic Arthritis
Cosentyx Gets First Us Approval In Children
The US Food and Drug Administration has approved Cosentyx for the treatment of moderate
To continue reading The Pharma Letter please , or claim a 7 day free trial subscription and access exclusive features, interviews, round-ups and commentary from the sharpest minds in the pharmaceutical and biotechnology space.
Or, if youre only interested in reading the content about a specific topic , then you can take our £10 per month channel subscription offer, which gives you access to all our news articles and in-depth content on this subject.
Can Using Cosentyx Make My Plaque Psoriasis Worse
No, Cosentyx shouldnt make your plaque psoriasis worse. If you believe your psoriasis is getting worse during your Cosentyx treatment, talk with your doctor. They may be able to determine whats causing your symptoms to worsen. Or they may recommend that you switch to a different medication to treat your condition.
Don’t Miss: What Is Plaque Psoriasis Look Like
Cosentyx Is A Biologic That Was Approved For The Treatment Of Psoriasis In 2015 And For The Treatment Of Psoriatic Arthritis In 2016
What is Cosentyx?
Cosentyx was approved by the FDA for the treatment of moderate-to-severe plaque psoriasis in adults in January 2015 and approved for use in children 6 years and older in June 2021. In January 2016, the FDA also approved Cosentyx to treat adults with active psoriatic arthritis.
For patients with plaque psoriasis, Cosentyx is administered by injection under the skin at weeks 0, 1, 2, 3, and 4 and then every 4 weeks. Health care providers may recommend a different dosing schedule for patients with psoriatic arthritis.
To learn more, please visit the Cosentyx website.
Tremfya Already Approved For Psoriasis Inhibits An Immune System Protein Called Il
Psoriatic arthritis may be treated with a wide variety of medications, from NSAIDs that focus on pain relief to disease-modifying drugs and biologics aimed at actually slowing the progression of this inflammatory condition. While many of the biologics used for psoriatic arthritis block an immune system protein called tumor necrosis factor , PsA patients have in recent years gained access to a growing number of drugs that target types of interleukin, another inflammatory protein.
In 2016, the FDA approved secukinumab , which targets IL-17A. In 2017, the agency approved ixekizumab , which also blocks IL-17A.
Now the FDA has approved a different interleukin blocker, but this one targets IL-23 instead.
Already approved for psoriasis, guselkumab is now the first IL-23 inhibitor to be FDA-approved for the treatment of psoriatic arthritis.
Another medication that targets both IL-23 and IL-12, ustekinumab has been approved to treat PsA since 2013.
Tremfya, which is taken via subcutaneous injection, can be used alone or along with a traditional DMARD such as methotrexate. According to Janssen, the drugs manufacturer, FDA approval for psoriatic arthritis was granted after two phase 3 clinical trials demonstrated that patients with active PsA who used it were more likely to have improvements in disease activity compared to those who took a placebo.
Whether or not Tremfya is more effective than other biologics for PsA is unclear.
Recommended Reading: Is Cheese Bad For Arthritis
Read Also: Best Shampoo For Psoriasis Canada
What Drugs Interact With Cosentyx
Cosentyx may interact with warfarin, cyclosporine, or live vaccines. Tell your doctor all medications and supplements you use and all vaccines you recently received. Tell your doctor if you are pregnant or plan to become pregnant before using Cosentyx. It is unknown if this drug passes into breast milk. Consult your doctor before breastfeeding.
Expand The Important Safety Information
References: 1. Taltz . Indianapolis, IN: Eli Lilly and Company 2021. 2. Callis Duffin K, Bagel J, Bukhalo M, et al. Phase 3, open-label, randomized study of the pharmacokinetics, efficacy and safety of ixekizumab following subcutaneous administration using a prefilled syringe or an autoinjector in patients with moderate-to-severe plaque psoriasis . J Eur Acad Dermatol Venereol. 2017 31:107-113.
INDICATIONS
Taltz is indicated for adults with active psoriatic arthritis , for adults with active ankylosing spondylitis , and for adults with active non-radiographic axial spondyloarthritis with objective signs of inflammation. Taltz is also indicated for patients aged 6 years or older with moderate-to-severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS Taltz is contraindicated in patients with a previous serious hypersensitivity reaction, such as anaphylaxis, to ixekizumab or to any of the excipients.
Pre-Treatment Evaluation for Tuberculosis Evaluate patients for tuberculosis infection prior to initiating treatment with Taltz. Do not administer to patients with active TB infection. Initiate treatment of latent TB prior to administering Taltz. Closely monitor patients receiving Taltz for signs and symptoms of active TB during and after treatment.
You May Like: What Is Arthropathic Psoriasis Unspecified
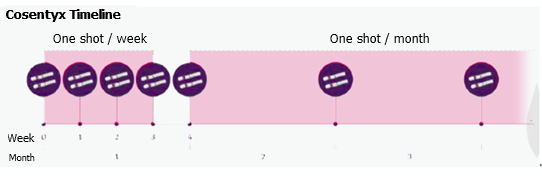
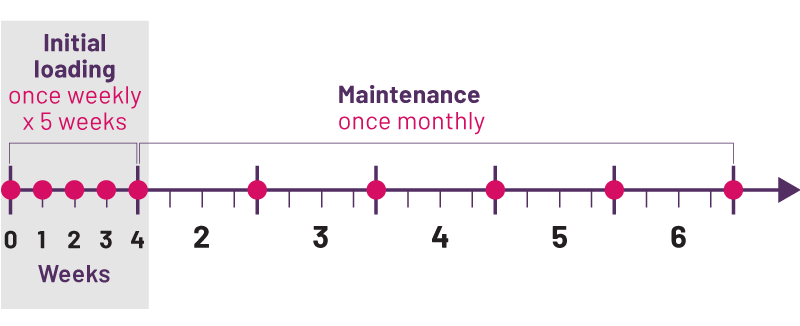
How Often Should I Use Cosentyx
At the start of your treatment with Cosentyx, youll need to get doses more often. This is to help the medication begin working more quickly in your body. When you first start your treatment, youll get a dose once per week for 5 weeks. After that, youll need a Cosentyx dose once every 4 weeks.
Here are some frequently asked questions about Cosentyx.
Trials 1 And : The Erasure And Fixture Studies
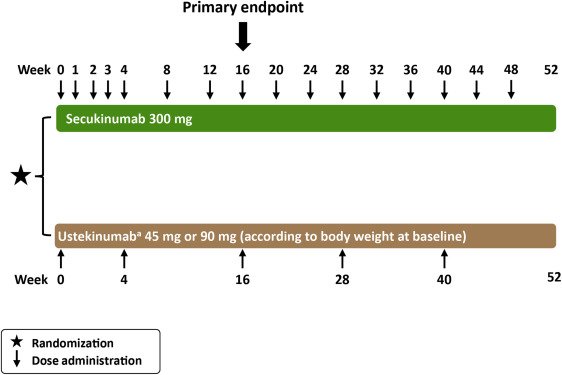
The ERASURE study was a 52-week, randomized, placebo-controlled trial that included 738 patients who received subcutaneous therapy at weeks 0, 1, 2, 3, and 4, followed by dosing every 4 weeks. Patients were randomized to secukinumab 300-mg or 150-mg doses for the first 4 weeks, followed by the same dose every 4 weeks, or to placebo.,
The FIXTURE study , also a 52-week, randomized, placebo-controlled trial, included 1306 patients 327 patients received secukinumab 300 mg, 327 patients received secukinumab 150 mg, 326 patients received placebo, and 323 patients received a biologic active control ., This discussion is limited to the data related to the secukinumab and the placebo groups.
The results of Trials 1 and 2 are shown in . In both studies, a greater proportion of patients who received secukinumab than those who received placebo or a biologic control met the PASI 75 criterion at week 12. At week 12 of Trial 1, a PASI 90 response was achieved in 59% of the secukinumab 300-mg group, 39% of the 150-mg group, and only 1% of the placebo group. At the same point in Trial 2, a PASI 90 response was achieved in 54% and 42% of the secukinumab groups versus 2% of the placebo group. The investigators concluded that the effectiveness of secukinumab in these trials validated IL-17A as a therapeutic target.
Dont Miss: How To Treat Arthritis In Lumbar Spine
Recommended Reading: How To Tell If You Have Psoriasis
Who Cosentyx Is Prescribed For
Cosentyx is approved to treat moderate to severe plaque psoriasis. The condition is considered moderate to severe if it covers 3% or more of your body. For reference, 1% of your body is about the size of your hand .
You must be able to have or systemic therapy. With phototherapy, light treats psoriasis plaques. Systemic therapy works in your whole body to stop the formation of plaques.
Cosentyx is also approved to treat active psoriatic arthritis. When the condition is active, you currently have symptoms.
Clinical trials have shown Cosentyx to be effective for treating psoriasis, including plaque psoriasis and psoriatic arthritis.
For treating plaque psoriasis, researchers looked at adults with moderate to severe plaque psoriasis. The researchers gave them either Cosentyx or a placebo . In 12 weeks, more adults who received Cosentyx reported that their symptoms eased than adults who received a placebo.
Researchers also compared Cosentyx with a placebo for treating adults with psoriatic arthritis. After 16 weeks, more adults who received Cosentyx reported that their symptoms eased than people who received a placebo.
Guidelines
Guidelines published by the American Academy of Dermatology recommend secukinumab, the active drug in Cosentyx, as a treatment option for adults with moderate to severe plaque psoriasis.
Choosing The Injection Sites
The injection sites are where the skin will be pierced to administer the subcutaneous injection.
The recommended site is the front of your thighs. You may also use the lower abdomen, but not the area five centimetres around the navel . If a caregiver is giving you the injection, the outer upper arms may also be used .
Choose a different site each time you give yourself an injection.
Do not inject into areas where the skin is tender, bruised, red, scaly or hard.
Avoid areas with scars or stretch marks
Don’t Miss: Psoriasis Area Severity Index Pasi
How To Use Cosentyx Syringe
Read the Medication Guide and the Instructions for Use leaflet provided by your pharmacist before you start using secukinumab and each time you get a refill. If you have any questions, ask your doctor or pharmacist.
Before treatment with this medication, your doctor will make sure that you have received all your vaccinations and also test you for tuberculosis . Your doctor should monitor you for symptoms of TB during and after treatment with this drug. If needed, treatment for tuberculosis or other infections should be given before receiving this medication.
This medication is given by injection under your skin as directed by your doctor. The injection is given in the upper arms, thighs, or abdomen . The dosage is based on your medical condition and response to treatment. For children, the dosage is also based on their weight. Follow your doctor’s instructions carefully.
Use this medication regularly to get the most benefit from it. It may help to mark your calendar with a reminder.
Tell your doctor if your condition does not improve or if it worsens.
What Is The Dosage For Cosentyx
Cosentyx is a medication thats injected under your skin. It comes in one strength, which is available in three forms. Two of these are forms that you can self-inject. The other form needs to be given by a healthcare provider.
When you first start Cosentyx, your doctor may give you your first dose at their office. They may also show you how to give yourself an injection. You can ask your doctor or pharmacist for tips on how to inject the drug so that you feel confident in the process.
Read Also: Are Tanning Beds Good For Psoriasis
What Are The Forms Of Cosentyx
Cosentyx comes in three different forms:
- Sensoready pen
- prefilled syringe
- single-use vial
The caps for the Sensoready pen and the prefilled syringe contain natural rubber. If you have a latex allergy, be sure to tell your doctor. A healthcare provider may need to give you Cosentyx. Depending on how severe your allergy is, your doctor may recommend another treatment.
Your doctor can help determine which form may be right for you.
What About Other Medications
Many patients need to continue another oral medication, like methotrexate or plaquenil, while on secukinumab, and should continue to take this if advised to do so by their doctor.Never take secukinumab with another any other biologic medication used to treat psoriatic arthritis, ankylosing spondylitis, or psoriasis.When you are taking secukinumab, it is very important that your doctors know if you are taking any other medicine. This includes prescription and non-prescription medicines as well as birth control pills, vitamins, and herbal supplements.
Don’t Miss: Best Soap For Eczema And Psoriasis
Preparation For Use Of Cosentyx Sensoready Pen And Prefilled Syringe
Before injection, remove COSENTYX Sensoready pen or COSENTYX prefilled syringe from the refrigerator and allow COSENTYX to reach room temperature without removing the needle cap.
The removable cap of the COSENTYX Sensoready pen and the COSENTYX prefilled syringe contain natural rubber latex and should not be handled by latex-sensitive individuals .
Inspect COSENTYX visually for particulate matter and discoloration prior to administration. COSENTYX injection is a clear to slightly opalescent, colorless to slightly yellow solution. Do not use if the liquid contains visible particles, is discolored or cloudy. COSENTYX does not contain preservatives therefore, administer the Sensoready pen or prefilled syringe within 1 hour after removal from the refrigerator. Discard any unused product remaining in the Sensoready pen or prefilled syringe.
If You Forget To Have It
If you have forgotten to inject a dose of COSENTYX, inject the next dose as soon as you remember. Then talk to your doctor to discuss when you should inject the next dose.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to have your medicine, ask your pharmacist for some hints.
Also Check: Psoriasis On Palms Of Hands Treatment
What Cosentyx Is Used For
COSENTYX is used for the treatment of the following inflammatory diseases:
- Plaque psoriasis
- Psoriatic arthritis
- Axial spondyloarthritis, including ankylosing spondylitis and non-radiographic axial spondyloarthritis
Plaque psoriasisCOSENTYX is used to treat a skin condition called ‘plaque psoriasis’ in adults.
Plaque psoriasis causes inflammation of the skin.
COSENTYX is used in adults with moderate to severe plaque psoriasis.
Psoriatic arthritisCOSENTYX is used to treat a condition called ‘psoriatic arthritis’. The condition is an inflammatory disease of the joints, often accompanied by psoriasis.
Axial spondyloarthritis, including ankylosing spondylitis and non-radiographic axial spondyloarthritis COSENTYX is used to treat a condition called ‘ankylosing spondylitis’ and ‘non-radiographic axial spondyloarthritis’ in adults. These conditions are inflammatory diseases primarily affecting the spine which causes inflammation of the spinal joints.
In patients with plaque psoriasis, psoriatic arthritis, and axial spondyloarthritis the body produces increased amounts of a protein called IL-17A. This may lead to symptoms such as itching, pain, scaling in psoriasis, swollen and tender joints in psoriatic arthritis, and pain in the spine in axial spondyloarthritis.
If you have any questions about how COSENTYX works or why this medicine has been prescribed for you, ask your doctor, pharmacist or healthcare provider.
COSENTYX may be used by people aged 65 years and over.
The Way Cosentyx Works
Cosentyx is classified as a monoclonal antibody. Monoclonal antibodies are proteins that work through your immune system, which is your bodys defense against infection.
Your immune system makes special types of proteins that can cause inflammation. Interleukin 17A is one of these proteins. Its been found to contribute to both plaque psoriasis and psoriatic arthritis. IL-17A can make symptoms of psoriasis worse. Researchers have found that people with plaque psoriasis and psoriatic arthritis have higher blood levels of IL-17A than people without these conditions.
The way Cosentyx works is to bind to IL-17A and prevent it from working. By preventing IL-17A from acting, Cosentyx helps decrease inflammation in your body. Inflammation leads to swelling and damage, which worsens plaque psoriasis and psoriatic arthritis. Because Cosentyx can prevent swelling and damage from occurring, the drug should help ease your symptoms.
Recommended Reading: How Can Psoriasis Be Treated
What If I Miss A Dose
If you miss your regular dose of Cosentyx, take it as soon as you remember. But dont double your normal Cosentyx dose. This could increase your risk for serious side effects.
If you miss a dose, call your doctor or pharmacist to find out when you should take your next dose.
If you need help remembering to take your dose of Cosentyx, try using a medication reminder. This can include setting an alarm, downloading a reminder app, or setting a timer on your phone. A kitchen timer can work, too.
You shouldnt use more Cosentyx than your doctor prescribes. Using more than this can lead to serious side effects.