Opportunityfor Adolescent Patients With Psoriasis To Take Part In A Trial Comparing Risankizumabwith Ustekinumab
This is an opportunityfor 12-17 year old patients with moderate to severe plaque psoriasis. Thetrial, sponsored by thepharmaceutical company, AbbVie, compares the treatment efficacy andsafety of Risankizumab vs Ustekinumab , two biologic therapies. All participants are guaranteed activetreatment, regardless of whether they have had systemic therapy before.
Recruitment criteria :
- Aged between 12 to < 18 years at the time ofenrolment
- Have had a diagnosis of chronic plaque psoriasisfor at least 6 months
- Have moderate to severe psoriasis, definedas 10% BodySurface Area psoriasis involvement with a static Physicians Global Assessment score of 3, or PsoriasisArea and Severity Index 12
- Erythrodermic psoriasis,generalised or localised pustular psoriasis, medication-induced or medicationexacerbated psoriasis, or new onset guttate psoriasis
- Active skin disease other than psoriasis that could interfere with theassessment of psoriasis
- Clinically significant drug or alcohol abuse within the last 6months
- An allergic reaction or hypersensitivity to a biologic agent orits excipients
- A latex allergy
- An organ transplant that requires continued immunosuppression
- Any malignancy exceptfor successfully treated non-melanoma skin cancer or localised carcinoma insitu of the cervix.
No history of:
No evidence of:
You can access the onlinesurvey here
For more information about thesurvey, please email Rachael Hewitt :
What Do Psoriasis Clinical Trials Look Like
The process that you will have to go through if you sign up for a clinical trial will be defined in precise terms before the recruitment. While there might be differences between various cases, participation in clinical trials usually involves these steps:
In some cases, Phase I trials require the volunteers to stay at the research facility, so medical professionals can monitor them around the clock.
A Study In Participants With Moderate To Severe Psoriasis
| The safety and scientific validity of this study is the responsibility of the study sponsor and investigators. Listing a study does not mean it has been evaluated by the U.S. Federal Government. Read our disclaimer for details. |
| First Posted : April 21, 2010Results First Posted : July 25, 2017Last Update Posted : September 24, 2019 |
The primary purpose for this study is to help answer the following research questions
- The safety of ixekizumab and any side effects that might be associated with it.
- Whether ixekizumab can help participants with Psoriasis.
- How much ixekizumab should be given to participants.
| Phase 2 |
| Study Type : |
| Quadruple |
| Primary Purpose: |
| A Dose-Ranging And Efficacy Study of LY2439821 In Patients With Moderate-To-Severe Psoriasis |
| Study Start Date : |
| Experimental: 10 mg Ixekizumab |
Part A:
10 milligrams ixekizumab given subcutaneous on weeks 0, 2, 4, 8, 12 and 16 for a total of six administrations.
Part B:
120 mg ixekizumab given SC Q4W. Subsequent to an amendment on May 2012, administration changed to 80 mg Q4W through Week 236.
Part C:
80 mg ixekizumab given SC Q4W through approximately week 344.
Part A:
25 mg ixekizumab given SC on weeks 0, 2, 4, 8, 12 and 16 for a total of six administrations.
Part B:
120 mg ixekizumab given SC . Subsequent to an amendment on May 2012, administration changed to 80 mg Q4W through Week 236.
Part C:
80 mg ixekizumab given SC Q4W through approximately week 344.
Part A:
Also Check: How To Cure Scalp Psoriasis Naturally
What Happens To Me And My Treatment Plan After A Clinical Trial
Your treatment plan after a clinical trial has ended will depend on your plan with your provider, your health insurance and access to health care.
If you are unable to access health care outside of a clinical trial, speak with the research staff about your treatment options after the trial. Or it may be possible to join another clinical trial.
Other Reasons People Choose To Participate
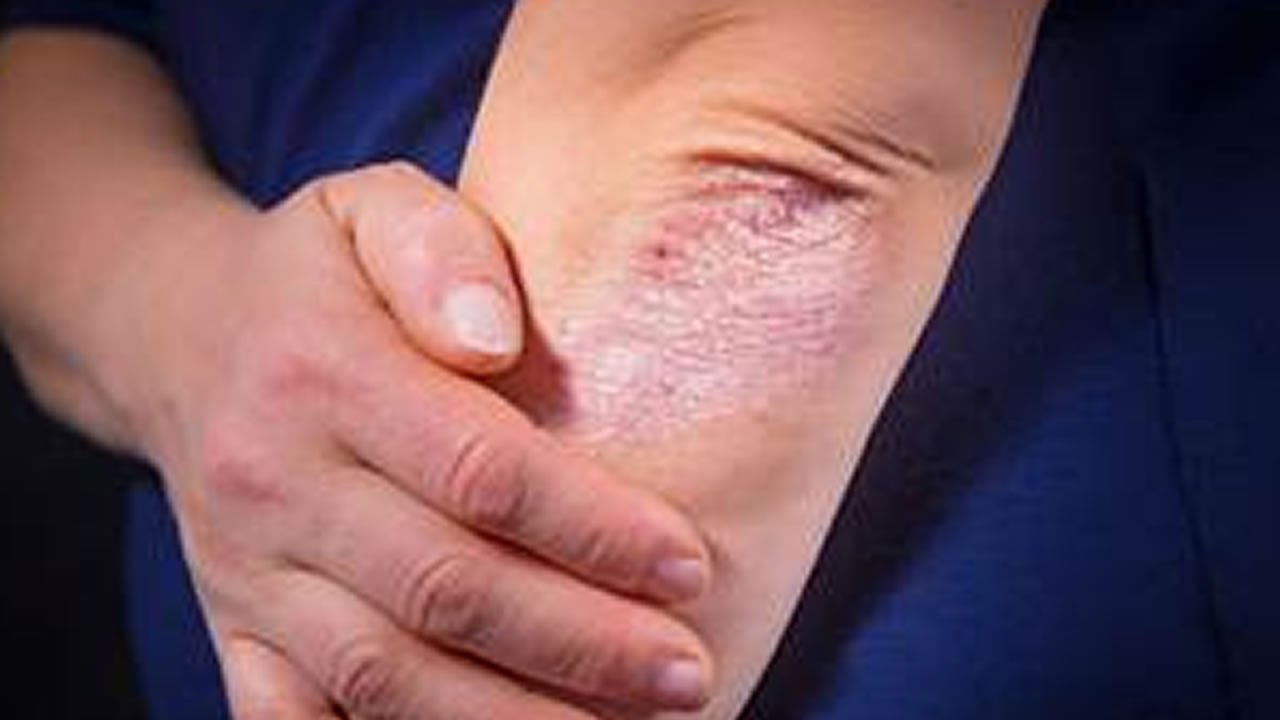
- You may benefit from new or better investigational medication not currently available to the public
- Any medical exams, tests, and/or procedures you receive during the study are provided at no cost to you
- You may receive additional care from healthcare providers
- You may also receive financial compensation for your time and travel
Don’t Miss: Psoriasis On Hands And Feet Treatment
Stelara And Tremfya Pregnancy Exposure Registry Otis Autoimmune Diseases In Pregnancy Project
open to eligible females
The purpose of the OTIS Autoimmune Diseases in Pregnancy Study is to monitor planned and unplanned pregnancies exposed to certain medications, to evaluate the possible teratogenic effect of these medications and to follow live born infants for one year after birth. With respect to fetal outcome, it is important to evaluate the spectrum of outcomes that may be relevant to a medication exposure during pregnancy, and these include both easily recognizable defects which are visible at birth, as well as more subtle or delayed defects that may not be readily identifiable without special expertise and observation beyond the newborn period.
La Jolla, California
Traditional Treatment Cautions Paid Clinical Trials For Psoriasis
You make an appointment to see your doctor because your psoriasis is flaring. He takes a look and then whips out his prescription pad to order one of the following:
. A prescription strength topical treatment
. phototherapy
. oral and injection therapy
. over the counter drugs
Any of these treatments may work fine to relieve your symptoms and make you feel better for now. The trouble with conventional treatment methods is that they rarely work to cure the underlying cause of this skin condition and that can leave you suffering repeat attacks. Still it is important to know what helps are available, and how they may help or hurt your chance of treatment success.
Topical Ointments Paid Clinical Trials for Psoriasis
Topical ointments are used at the first sign of psoriasis. If caught early enough they can sometimes stave off a full-blown attack. Unfortunately, most people report becoming immune to them when used often, which makes them useless for long term use. Still, using the occasional cream or gel can help relieve some of your symptoms. Steroids
Both topical and internal steroids can be prescribed to help ease psoriasis swelling inflammation and itching. Immunomodulators
An immune suppressing drug that was originally used to help transplant patients beat organ rejection, Cyclosporine helps to inhibit T lymphocyte production so that scaly plaque is unable to grow on the surface of the skin.
Oral Retinoid Paid Clinical Trials for Psoriasis
. stretch marks
Don’t Miss: How Much Epsom Salt In Bath For Psoriasis
Types Of Clinical Trials
Clinical trials may be divided into types, or phases. Each new IMP must proceed through the phases in turn. Unless they pass each phase, they cannot proceed to the next.
Phase one:Phase one clinical trials are the first time that a potential new medicine or other intervention is given to a human being. This is done using a small number of healthy volunteers. The main aim of a phase one trial is to check that there are no serious side effects, known as adverse events, with the planned dose of the potential medicine. Phase one studies go ahead with extreme caution to prevent multiple side effects each volunteer is closely monitored by doctors and research staff. The trial starts by giving healthy volunteers very small doses of the potential new medicine, then increasing the dose to check what doses are acceptably safe.
Phase three:A phase three study tests the medicine on a larger group of patients. In phase three trials, patients treated with the new product may have other medical problems, rather than sharing similar characteristics as in a phase two trial. This is to ensure that all types of patients can be successfully and safely treated with the new medicine. Often, trial populations are not truly representative of the general psoriasis or psoriatic arthritis population.
At the end of phase three, the information needed for the product information leaflet and drug label will be completed.
Donotpay Finds The Best Psoriasis Clinical Trials For You
Psoriasis is an autoimmune disease that usually appears as red, dry, and itchy patches of skin. Around 2-3% of people worldwide suffer from this condition that doesnt have a cure.
While the affected population has multiple treatment options to keep the disease in check, more and more clinical trials are trying to find the definitive cure or new ways to improve the life quality of patients with psoriasis.
Also Check: How To Diagnose Scalp Psoriasis
Grade Recommendations For Therapies
Each of the six domain groups and the related conditions group synthesized the evidence they extracted from the literature reviews and developed recommendations by consensus. A summary of these recommendations is given in Table . Using the GRADE approach resulted in strong or conditional recommendations either for or against different therapies, or a decision that no recommendation could be made if there was insufficient or contrasting evidence. Recommendations regarding each domain were presented for discussion in the wider group, and minor changes were made for consistency of interpretation across the groups. Wherever possible, drugs were grouped by class unless there was strong evidence of differences in efficacy within the class. The summary table shows the recommendation for each drug class within the different domains of PsA and related conditions but does not provide a hierarchy within the strong or conditional recommendations, as these nuances are addressed in the text.
Table 3 Summary of recommendations for treatment of PsA
New Clinical Trials For Psoriasis
Although psoriasis has been acknowledged far back in history, its cause is not 100% understood. In moderate to severe cases psoriasis can greatly lower the patients quality of life. They may not be able to participate in sports, or work in certain industries due to their psoriasis.
It is important that we continue to conduct psoriasis clinical research. In recent years there have been several psoriasis clinical trials that show promise in better treating this disease. Hopefully it will only be a matter of years before an economical, safe and effective psoriasis treatment is readily available.
Read Also: Scalp Psoriasis Cause Hair Loss
Does Pcos Cause A Weak Immune System
PCOS may overstimulate the immune system and may result in the overproduction of antibodies. Health experts label PCOS as an autoimmune disease and treat it as such. For some PCOS patients, joining a clinical trial could be their best shot. These clinical trials offer potential drugs that may cure PCOS, but one must pass the eligibility requirements. If you are interested in participating in a PCOS clinical trial, you may quickly find one by using the Power website.
What If I Am Not Eligible To Participate In A Clinical Trial But I Want To Contribute To Research

There are many ways to contribute to scientific research.
- The National Psoriasis Foundation and CorEvitas, LLC have developed the CorEvitas Psoriasis Registry, which helps to advance medical research and improve patient care. Joining this registry will help researchers better understand the safety and efficacy of psoriasis treatments, comorbidities of psoriasis and the natural history or progression of psoriasis.
- Citizen Pscientist is the first-ever global online research network designed to increase knowledge and give researchers new information and leads. You get to evaluate the information and data gathered and discuss topics that you find interesting.
- Donate to Research and help support projects on the leading edge of research into psoriasis and psoriatic arthritis including research aimed at accelerating the discovery, development and delivery of new treatments.
- Join our team of advocates to urge your lawmakers to increase federal funding for psoriatic disease research.
- Patient-Centered Research places the you at the center of the research process. You can be involved throughout the research process, from participating in patient surveys, to the formulation of research questions, to the dissemination of results.
Read Also: Psoriasis Scalp Shampoo Natural Remedies
> > > Best Psoriasis Cure Available
3. To help safeguard the skin, the immune system release chemicals that can cause nerves to itch and blood vessels to dilate to prepare the skin for a sudden rush of immune cells
4. When the immune cells arrive at the scene, most work to kill off the pathogens causing the distress, but a few capture some of the invaders and take them back to the heart of the immune system, where other soldier cells are produced in a way to recognize and attack the invading cells on contact
5. Inflammation is the bodys way of opening blood vessels to allow more soldier cells to rush to the battlefield.
While this entire process is completely normal, people with psoriasis tend to overproduce these soldier cells when the body feels threatened. This overabundance of killer immune cells can actually be dangerous to the skin since they begin to attack good cells along with the bad ones.
While it is great to finally understand the impact an improperly working immune system can have on your skin and cause psoriasis more research is needed to pinpoint the exact cause for the over-firing of the cell messages. Paid Clinical Trials for Psoriasis
Team Clara Interview With Vickie
The team at Clara has gotten in touch with Vickie, who is a psoriasis patient. In this interview, Vickie shares her experience living with the disease, as well going through psoriasis clinical trials.
Lilly:
Vickie, I’d love to chat with you a little bit more about if youve been in just one clinical trial or in multiple trials?
Vickie:
I’ve only been in one.
Lilly:
Okay. And was it for your psoriasis?
Vickie:
Yes, it was. It was a pill. I don’t have the paper in front of me, but I can email you later on to tell you exactly what it was. But it was a pill form that you took twice a day, one in the morning, one at night. It was in phase two, so there was no placebo. It was the straight medication.
Lilly:
Oh, that’s great that nobody got the placebo.
Vickie:
No. No, nobody got the placebo. And the remarkable thing for me is, I basically noticed a difference right away.
Lilly:
And did you have in person appointments for the trial?
Vickie:
Oh yeah, it was in person. The thing is I live in Shreveport, Louisiana, and the clinical trial, which is the closest one I could find, was in Dallas.
Lilly:
Oh, wow. Did you drive to Dallas?
Vickie:
Yeah, it was in Dallas. So I had to drive three hours one way to get to the clinical trial.
Lilly:
Wow. And how did you hear about the clinical trial?
Vickie:
Lilly:
Yeah. And did you say you weren’t being treated before the clinical trial?
Vickie:
Lilly:
Hmm.
Vickie:
Lilly:
And that’s all they wanted to treat you with.
Vickie:
Lilly:
Vickie:
Don’t Miss: Are You Born With Psoriasis
The Informed Consent Provides You With Information Related Specifically To The Trial You Are Participating In So You Will Have A Clear Understanding Of The Study Your Role And The Expectations Of You Before You Begin
Giving written consent to participate in a clinical trial means that you acknowledge, understand and accept all aspects of the study. This includes any possible benefits or risks. The physicians and research staff are responsible for helping you understand the information. They should give you enough time to make a decision about participating in the clinical trial. You can always ask questions if you do not understand something about the study or if you have other concerns.
Dermatologic Research Evaluation And Monitoring Of Sleep In Moderate
open to eligible people ages 18 years and up
Getting enough sleep is important for maximizing health and well-being. When it comes to health, sleep is as vital as regular exercise and eating a balanced diet. Not getting enough sleep can lead to health problems like heart attacks, diabetes, and even cancer. Since individuals with psoriasis have these same health problems, getting better sleep may help to keep them happier and healthier. This study will look at how individuals with psoriasis sleep and if their sleep is different than indivuduals without psoriasis.
San Francisco, California
Recommended Reading: Nail Psoriasis Over The Counter Treatment
Treatments That Become Approved May Continue To Have Clinical Studies Done In Order To:
- Compare the safety, effectiveness and cost of the new treatment with other treatments that are currently available
- Better understand the long-term safety and efficacy of the new treatment
- Measure its impact on quality of life for people using the new treatment
- Evaluate the treatment in other types of patients
- To further characterize the treatment
What Tests Need To Be Done For Pcos
There are several tests that your doctor must ask you to undergo to confirm since there is no single test that can determine whether you have PCOS or not. Aside from the confirmation that you have PCOS, your doctors must also understand your symptoms to ensure that they recommend the best course of treatment. They may also recommend you to participate in some clinical trials for potential cures which are under clinical trials. If you want to look for a clinical trial that fits your overall health condition and within your area, please visit Power.
Also Check: Home Remedies For Psoriasis Of The Skin
Making An Educated Decision
People join clinical trials for many reasons. Some people may be looking to take on a more active role in their healthcare, while others may be more interested in helping future generations. No matter the reason, its important that you make an educated decision.
You and your health care provider should discuss if clinical trials are right for you. When making any type of treatment decision, you might consider your disease severity, your reactions to previous treatments and other health conditions you might be coping with.
Keep in mind that you may withdraw from a study at any time for any reason.